Transformations in Matter and Energy Carbon TIME is an NSF-funded partnership led by Michigan State University
Plants | Lesson 5 - Explaining How Plants Grow
Students develop a story about how the high-energy glucose molecules created during photosynthesis are transformed into larger organic polymers during biosynthesis in plants.
Guiding Question
How can a potato plant make a potato?
Activities in this Lesson
Note: There are multiple pathways to choose from in Lessons 4-5. Please see the Plants Unit Read Me document, the Student Challenges and Teacher Choices in the Plants Unit document, and/or the Background Information section below for clarification in making this instructional decision.
- Activity 5.1: Tracing the Process of Potatoes Growing: Biosynthesis (40 min)
-

 Activity 5.2: Explaining How Plants Grow: Biosynthesis (40 min)
Activity 5.2: Explaining How Plants Grow: Biosynthesis (40 min) - Activity 5.3: Explaining How Plants Grow: Biosynthesis (40 min)
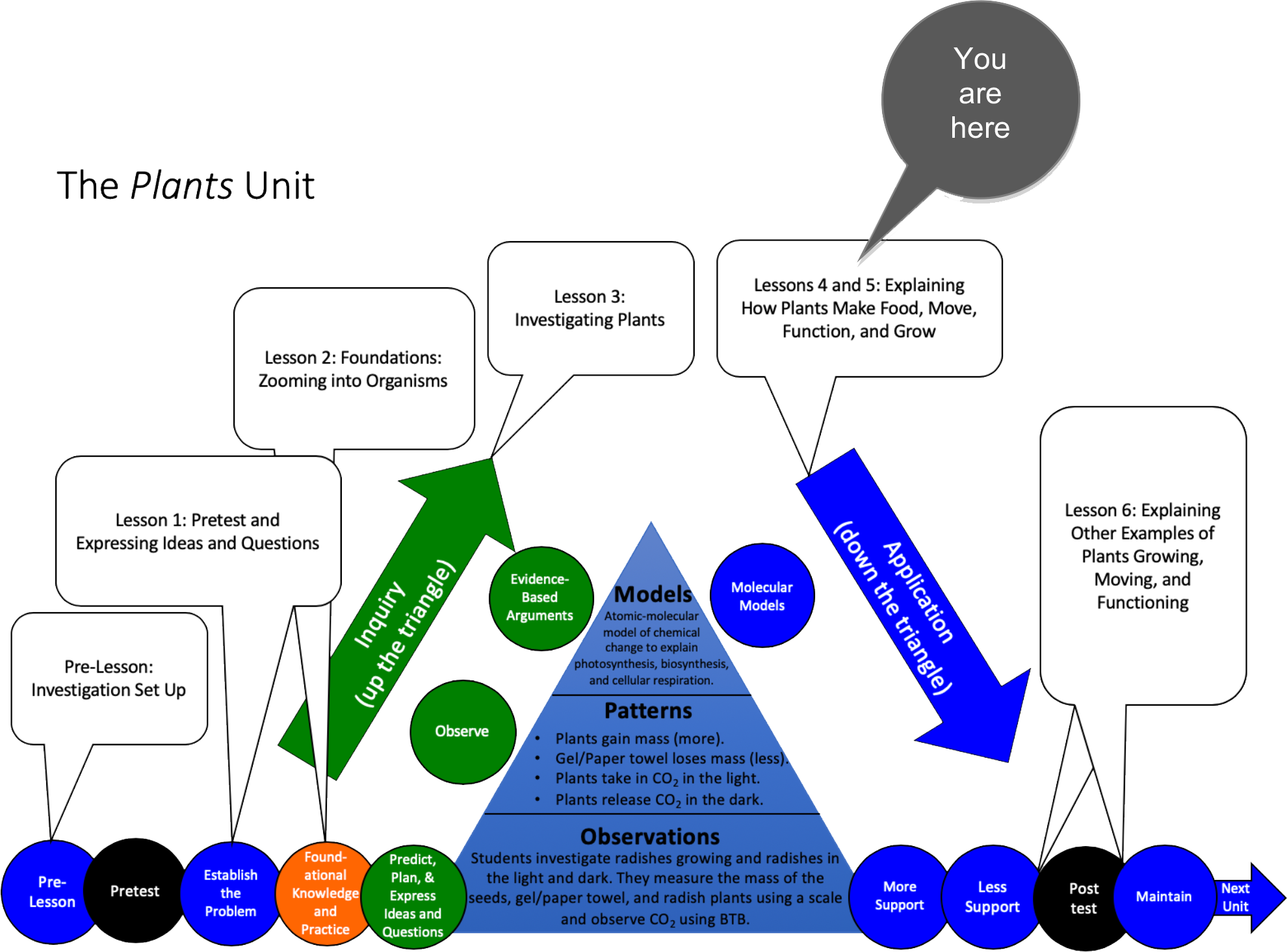
Unit Map
Target Performances
|
Activity |
Target Performance |
|
Lesson 5 – Explaining How Plants Grow (students as explainers) |
|
|---|---|
|
Activity 5.1: Tracing the Process of Potatoes Growing: Biosynthesis |
Students “zoom in” to the structure and function of a potato plant’s systems and cells, tracing atoms and energy. |
|
Optional Activity 5.2: Molecular Models for Potatoes Growing: Biosynthesis |
Students use molecular models to explain how plants make monomers from glucose and minerals and monomers are linked into polymers during biosynthesis. |
|
Activity 5.3: Explaining How Potato Plants Grow: Biosynthesis |
Students explain how matter moves and changes and how energy changes during biosynthesis in a potato plant’s cells. |
NGSS Performance Expectations
Middle School
- MS. Structure and Properties of Matter. MS-PS1-1. Develop models to describe the atomic composition of simple molecules and extended structures.
- MS. Chemical Reactions. MS-PS1-2. Analyze and interpret data on the properties of substances before and after the substances interact to determine if a chemical reaction has occurred.
- MS. Chemical Reactions. MS-PS1-5. Develop and use a model to describe how the total number of atoms does not change in a chemical reaction and thus mass is conserved.
- MS. Structure, Function, and Information Processing. MS-LS1-3. Use argument supported by evidence for how the body is a system of interacting subsystems composed of groups of cells.
- MS. Matter and Energy in Organisms and Ecosystems. MS-LS1-7. Develop a model to describe how food is rearranged through chemical reactions forming new molecules that support growth and/or release energy as this matter moves through an organism.
- MS. Matter and Energy in Organisms and Ecosystems. MS-LS2-3. Develop a model to describe the cycling of matter and flow of energy among living and non-living parts of an ecosystem.
High School
- HS. Matter and its Interactions. HS-PS1-4. Develop a model to illustrate that the release or absorption of energy from a chemical reaction system depends upon the changes in total bond energy.
- HS. Chemical Reactions. HS-PS1-7. Use mathematical representations to support the claim that atoms, and therefore mass, are conserved during a chemical reaction.
- HS. From Molecules to Organisms: Structures and Processes. HS-LS1-2. Develop and use a model to illustrate the hierarchical organization of interacting systems that provide specific functions within multicellular organisms.
- HS. Matter and Energy in Organisms and Ecosystems. HS-LS1-6. Construct and revise an explanation based on evidence for how carbon, hydrogen, and oxygen from sugar molecules may combine with other elements to form amino acids and/or other large carbon-based molecules.
Talk and Writing
At this stage in the unit, the students will complete the inquiry and application sequences for Plant Investigations—they will go up and down the triangle. This means they will go through the Observations Phase, the Evidence-Based Arguments Phase, and the Explanations Phase. The tables below show specific talk and writing goals for these phases of the unit.
|
Talk and Writing Goals for the Observations Phase |
Teacher Talk Strategies That Support This Goal |
Curriculum Components That Support This Goal |
|---|---|---|
|
Help students discuss data and identify patterns. |
What patterns do we see in our data? How do you know that is a pattern? What about ______ data. What does this mean? |
Class Results Poster Class Results Spreadsheet |
|
Encourage students to compare their own conclusions about the data and evidence with other groups and other classes. |
What about this number? What does this tell us? How is group A’s evidence different from Group B’s data? How do our class’s data differ from another classes’ data? |
Class Results Spreadsheet Class Results Poster Investigation Video (selected segments) |
|
Make connections between the observations and the data/evidence. |
It says here that our BTB turned colors. What does that mean? You recorded that your plant gained mass. What does that mean? |
|
|
Have students consider how their predictions and results compare. |
Let’s revisit our predictions. Who can explain the difference between our class predictions and our results? Who had predictions that were similar to our results? Has your explanation changed? How? |
|
|
Talk and Writing Goals for the Explanations Phase |
Teacher Talk Strategies That Support This Goal |
Curriculum Components That Support This Goal |
|---|---|---|
|
Examine student ideas and correct them when there are problems. It’s ok to give the answers away during this phase! Help students practice using precise language to describe matter and energy. |
Let’s think about what you just said: air molecules. What are air molecules? Are you talking about matter or energy? Remember: atoms can’t be created. So that matter must have come from somewhere. Where did it come from? Let’s look at the molecule poster again… is carbon an atom or a molecule? |
Molecule Poster Three Questions Poster
|
|
Focus on making sure that explanations include multiple scales. |
The investigation gave us evidence for what was happening to matter and energy at a macroscopic sale. But what is happening at an atomic-molecular scale? What is happening to molecules and atoms? How does energy interact with atoms and molecules during chemical change? Why doesn’t the macroscopic investigation tell us the whole story? Let’s revisit our scale poster… what is happening to matter at the molecular scale? |
Molecular Models Molecular Modeling Worksheets Explanations Tool PPT Animation of chemical change Powers of Ten Poster |
|
Encourage students to recall the investigation. |
When did this chemical change happen during our investigation? How do we know that? What is our evidence? What were the macroscopic indicators that this chemical change took place? |
Evidence-Based Arguments Tool Investigation Video |
|
Elicit a range of student explanations. Press for details. Encourage students to examine, compare, and contrast their explanations with others’. |
Who can add to that explanation? What do you mean by _____? Say more. So I think you said _____. Is that right? Who has a different explanation? How are those explanations similar/different? Who can rephrase ________’s explanation? |
Explanations Tool |

 Download PDF of Lesson 5 Teacher's Guide
Download PDF of Lesson 5 Teacher's Guide